AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Lithium atom model12/2/2023  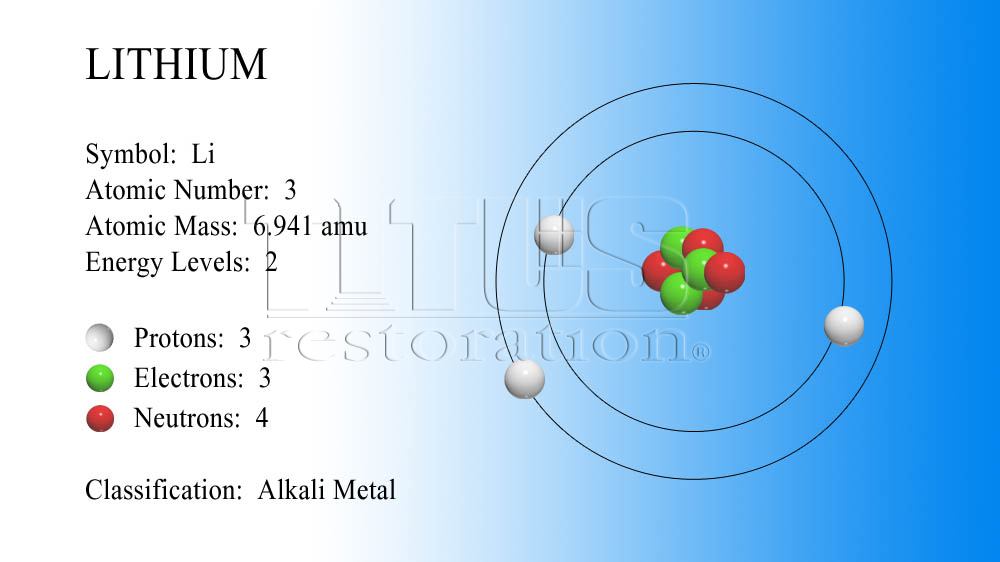
The 3 d orbital is higher in energy than the 4 s orbital. However, this pattern does not hold for larger atoms. The energy increases as we move up to the 2 s and then 2 p, 3 s, and 3 p orbitals, showing that the increasing n value has more influence on energy than the increasing l value for small atoms. The 1 s orbital at the bottom of the diagram is the orbital with electrons of lowest energy. Figure 6.24 depicts how these two trends in increasing energy relate. In any atom with two or more electrons, the repulsion between the electrons makes energies of subshells with different values of l differ so that the energy of the orbitals increases within a shell in the order s < p < d < f. The energy of atomic orbitals increases as the principal quantum number, n, increases. The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. This allows us to determine which orbitals are occupied by electrons in each atom. Having introduced the basics of atomic structure and quantum mechanics, we can use our understanding of quantum numbers to determine how atomic orbitals relate to one another. Relate electron configurations to element classifications in the periodic table.Identify and explain exceptions to predicted electron configurations for atoms and ions.Derive the predicted ground-state electron configurations of atoms.The Bohr model is the one most often depicted when drawing an atom.By the end of this section, you will be able to:.Niels Bohr helped rescue and provide jobs for scientists escaping Germany during the Nazi regime by giving them positions at the theoretical physics institute he ran and helping them get visas to other countries.In his later years, Niels Bohr advocated for openness between nations in atomic weapons development.Niels Bohr was awarded the Nobel Prize in physics in 1922 for his work investigating the structure of an atom.Does not match what scientists would later learn that an electron can be both a wave and a particle.The Bohr model did not describe the changes seen in emission spectra when a magnetic field was present (known as the Zeeman effect).The atomic model could not explain the different line intensities in emission spectra.The emission spectrum of atoms with more than one electron could not be explained. The Bohr atomic model could not accurately describe larger atoms. 
Hydrogen and other 1 electron systems are the only ones accurately explained by the Bohr Model Problems with Bohr’s Model When there is more than one electron interactions between the nucleus and electrons become too complicated for the Bohr model.ĭepiction of the Bohr model of hydrogen. Other ions that also have one electron can also be explained accurately (for example, He +). When there is more than one electron the model does not accurately predict the energies. The Bohr model of hydrogen is the only one that accurately predicts all the electron energies. Previous models had not been able to explain the spectra. Using Bohr’s model of the atom the previously observed atomic line spectrum for hydrogen could be explained. No other model had done this before and was a big step towards the development of quantum mechanics. It is the first atom model that accounts for quantized or discrete energy steps. Thomson in 1904), the Saturnian model (by Hantaro Nagaoka in 1904), and the Rutherford model (by Ernest Rutherford in 1911).īohr’s model is different from the preceding model (the Rutherford model) because electrons can only orbit at certain radii or energy. The Bohr model replaced earlier models such as the plum-pudding model (by J.J. (From Wikipedia Commons) Improvements From Previous Models 
Each orbit change has a unique energy difference.Ītomic line spectra of hydrogen. And the blue line would be caused by an electron moving from shell 3 to shell 2. For example, the red line would be caused by the electron moving from shell 2 to shell 1. Only light of specific energy (or color) is released, shown by the sharp lines seen in the spectra, not all colors of light. These discrete energy steps are what cause atomic line spectra, like the one seen for hydrogen below. The energy is released in the form of light. When the electron moves from a larger higher-energy shell to a smaller lower-energy one it releases energy. When an electron moves to a smaller shell, it releases energy which we observe as light. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
